 Both a magnetic (dipole) moments will interact with each other via spin-orbit coupling to generate fine structure atomic spectra. 
When hydrogen gas is placed into a tube and electric current passed through it, the color of emitted light is pink. Similarly, the rotating electron (really intrinsic spin) of the electron induces a different magnet. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. Using this table we can pick out the possible electronic states easily since all terms are concentric rectangles on the table. Figure 8.11.1 : An electron that orbits the nucleus induces a 'current' and a magnetic field. 6 Each row of the table represents a total magnetic quantum number, while each column does a total spin. 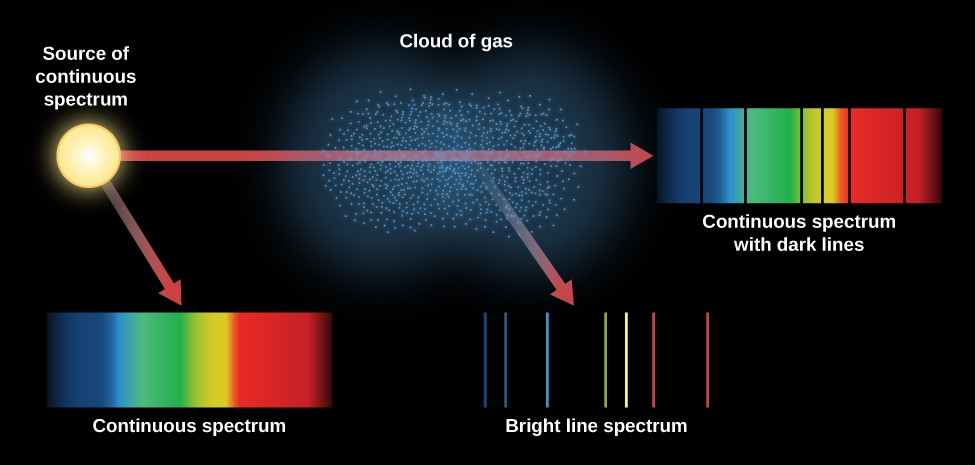
It requires a table of possibilities of different "micro states," which happened to be called "Slater's table". Atomic absorption spectroscopy is based on absorption of light by free metallic ions. Here is a commonly used method to determine term symbols for an electron configuration. Atomic absorption spectroscopy ( AAS) and atomic emission spectroscopy ( AES) is a spectroanalytical procedure for the quantitative determination of chemical elements by free atoms in the gaseous state. Selection rules have been derived for electromagnetic transitions in molecules, in atoms, in atomic nuclei, and so on. Such as 3 D 2, where S = 1, L = 2, and J = 2. In physics and chemistry, a selection rule, or transition rule, formally constrains the possible transitions of a system from one quantum state to another. Term symbols usually represent electronic states in the Russell-Saunders coupling scheme, where a typical atomic term symbol consists of the spin multiplicity, the symmetry label and the total angular momentum of the atom. Term symbols for an Electron Configuration A spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum, resulting from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. The absorption spectrum of a chemical element or chemical compound is the spectrum of frequencies or wavelengths of incident radiation that are absorbed by the compound due to electron transitions from a lower to a higher energy state. 
Supposing the gyromagnetic ratio of an electron is 2. \): When light from a hydrogen gas discharge tube is passed through a prism, the light is split into four visible lines.\] 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
